AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Density of water slugft33/21/2024 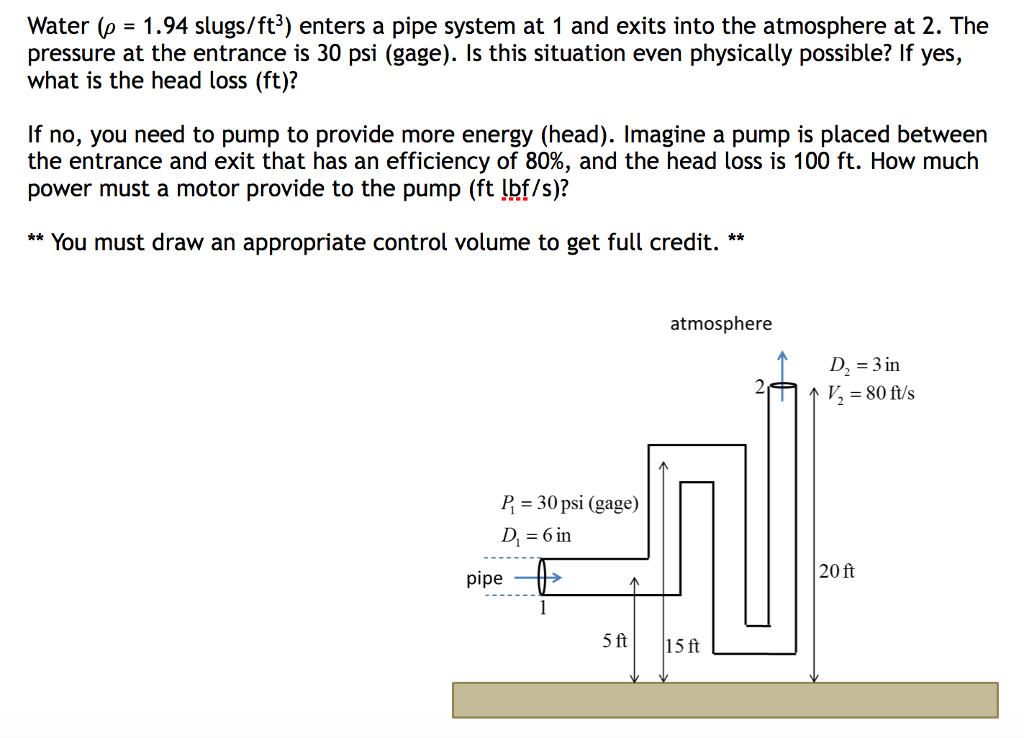
Weight or density of ditch filling materials.ĭry Air and Water Vapor - Density and Specific Volume vs. Specific Weight and Specific GravityĪn introduction to density, specific weight and specific gravity.ĭensities - pound per cubic foot and kilograms per cubic metre - of dirt and mud. Density of inorganic substances in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensity vs. Density of acetic acid, citric acid, formic acid, D-lactic acid, oxalic acid and trichloroacetic acid in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensities of common products - Imperial and SI-units.ĭensity of Aqueous Solutions of some Inorganic SubstancesĬhanges in density of aqueous solutions with changes in concentration at 20☌. Density of inorganic sodium salts in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensities of Aqueous Solutions of Organic AcidsĬhanges in density of aqueous solutions with changes in concentration at 20☌. Density of potassium salts in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensities of Aqueous Solutions of Inorganic Sodium SaltsĬhanges in density of aqueous solutions with changes in concentration at 20☌. Viscosity at 20☌/68☏ and 50☌/122☏ for more than 120 crudes is shown as function of specific of Aqueous Solutions of Inorganic Potassium SaltsĬhanges in density of aqueous solutions with changes in concentration at 20☌. 
Variations in crude oil density are shown as function of temperatur, together with volume correction factors. Online calculators, figures and tables showing density and specific weight of liquid and gaseous butane, C 4H 10, at varying temperarure and pressure, SI and Imperial units.Ĭalculator and formulas for conversion between different units of concentration: Molarity, molality, mole fraction, weight percent of solute and grams of solute per liter of solution - descriptive terms for solubility. Online calculator, figures and table showing density and specific weight of benzene, C 6H 6, at temperatures ranging from 5 to 325 ☌ (42 to 620 ☏) at atmospheric and higher pressure - Imperial and SI Units.īutane - Density and Specific Weight vs. Online calculator, figures and tables showing density and specific weight of argon, Ar, at varying temperature and pressure - Imperial and SI Units.īenzene - Density and Specific Weight vs. Online calculator with figures and tables showing density and specific weight of ammonia for temperatures ranging -50 to 425 ☌ (-50 to 800 ☏) at atmospheric and higher pressure - Imperial and SI Units. An ammonia phase diagram are included.Īmmonia - Vapour Pressure at Gas-Liquid Equilibriumįigures and table with ammonia saturation pressure at boiling points, SI and Imperial units.Īmmonia Gas - Density vs. 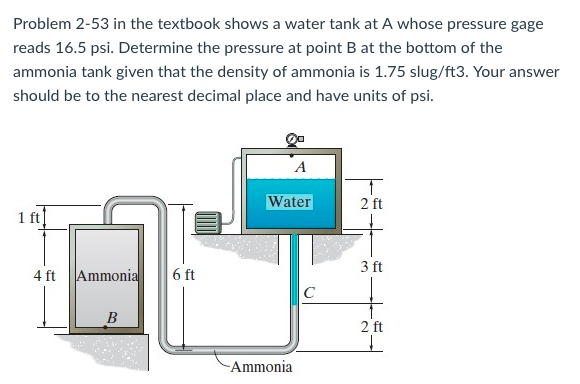
Molweight, melting and boiling point, density, pKa-values, as well as number of carbon and hydrogen atoms in molecules are given for 150 different alcohols and acids.Īmmonia - Properties at Gas-Liquid Equilibrium Conditionsįigures and tables showing how the properties of liquid and gaseous ammonia changes along the boiling/condensation curve (temperature and pressure between triple point and critical point conditions). Online calculator, figures and tables showing density and specific weight of acetone at temperatures ranging from -95 to 275 ☌ (-138 to 530 ☏) at atmospheric and higher pressure - Imperial and SI Units.Īlcohols and Carboxylic Acids - Physical Data Material properties of gases, fluids and solids - densities, specific heats, viscosities and more. long ton/yd 3 - long ton (=imperial ton) per cubic yardĭensities of solids, liquids and gases.lb/gal (US liq) - pound per US liquid gallon.lb/gal (Imp) - pound per imperial gallon.oz/gal (US liq) - ounce per US liquid gallon.oz/gal (Imp) - ounce per imperial gallon.There are exceptions however, such as water's density increasing between 0☌ and 4☌.īelow is a table of units in which density is commonly expressed, as well as the densities of some common materials. Increases in temperature tend to decrease density since the volume will generally increase. An increase in pressure decreases volume, and always increases density. However, when regarding gases, density is largely affected by temperature and pressure. In the case of solids and liquids, the change in density is typically low. Note that density is also affected by pressure and temperature. It is useful to carefully write out whatever values are being worked with, including units, and perform dimensional analysis to ensure that the final result has units of mass There are many different ways to express density, and not using or converting into the proper units will result in an incorrect value. However, it is important to pay special attention to the units used for density calculations. The calculation of density is quite straightforward.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
